 RSS
RSS
Press Releases
Trusted reliable news sources from around the web. We offer special news reports, topic news videos, and related content stories. Truly a birds eye view on news.
- Details
- Written by Send2Press NewsWire
- Category: Send2Press NewsWire Distribution Service
TECUMSEH, Mich., June 10, 2026 (SEND2PRESS NEWSWIRE) -- The parent company of DiggyPOD, Everbrook Holdings, announced the acquisition of Long Overdue Books, a Chicago-area publishing company known for guiding authors from first draft to finished book. The deal closed on May 15, 2026, and brings Long Overdue Books into the Everbrook family alongside DiggyPOD. DiggyPOD is a Tecumseh, Michigan-based book printer that became Everbrook's first portfolio company in June 2025.


- Details
- Written by Send2Press NewsWire
- Category: Send2Press NewsWire Distribution Service
NEW YORK, N.Y., June 10, 2026 (SEND2PRESS NEWSWIRE) -- As conversations around declining fertility rates and reproductive health continue to gain global attention, the World Fertility Project (WFP), produced by pregnantish, today announced the return of its international "Break the Taboo" campaign, launching June 17 in honor of World Infertility Awareness Month.


- Details
- Written by Send2Press NewsWire
- Category: Send2Press NewsWire Distribution Service
CHICO, Calif., June 10, 2026 (SEND2PRESS NEWSWIRE) -- With West Coast architectural fees climbing to historic highs, standard residential design services are rapidly becoming cost-prohibitive for middle-class homebuilders. In a direct bid to dismantle this financial bottleneck, California-based manufacturer Sierra Log and Timber today announced the rollout of a flat-rate $950 custom 3D architectural drafting program.


- Details
- Written by Send2Press NewsWire
- Category: Send2Press NewsWire Distribution Service
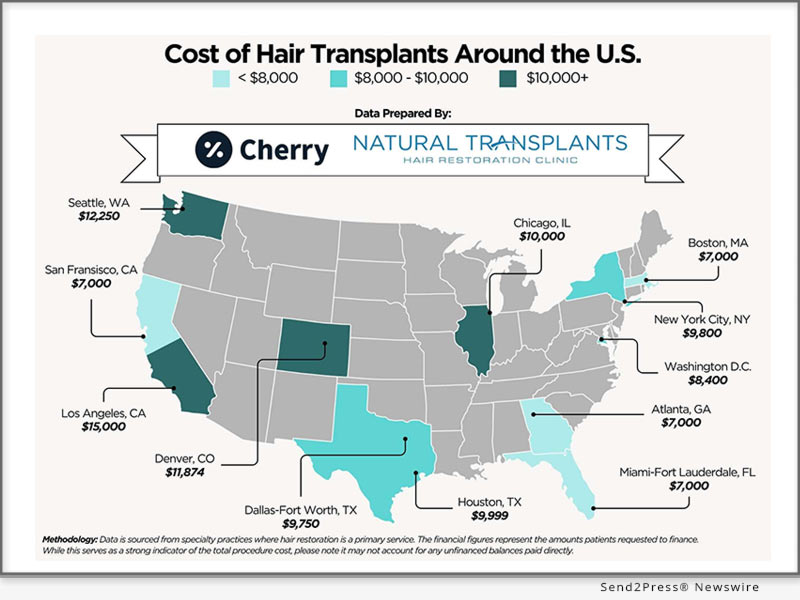
FORT LAUDERDALE, Fla., June 10, 2026 (SEND2PRESS NEWSWIRE) -- Natural Transplants, a leader in physician-led hair restoration, and Cherry, a leading patient financing platform, today announced a strategic partnership aimed at improving access to high-quality hair transplant procedures nationwide. Alongside the partnership, the companies released an exclusive 2026 guide to hair transplant cost in the U.S., offering new insight into the significant cost disparities patients face across major metropolitan markets.

- Details
- Written by Globe NewsWire Press Releases
- Category: Globe NewsWire News Distribution Service
Selskabsmeddelelse nr. / Corporate Announcement no - 34 / 2026 10. juni 2026 Gennemførelse af kapitalforhøjelse, nom. kr. 321.800 / Implementation of capital increase, nom. DKK 321.800 Bestyrelsen i NewCap Holding A/S har i henhold til den i vedtægterne angivne bemyndigelse d.d. besluttet at forhøje selskabskapitalen med nom. DKK 321.800. Den gennemførte kapitalforhøjelse sker til…
- Details
- Written by Globe NewsWire Press Releases
- Category: Globe NewsWire News Distribution Service
Company Announcement No 30/2026 Tel +45 74 37 37 37 AL Sydbank A/SCVR No DK 12626509, Aabenraaal-sydbank.com Dear Sirs Transactions with shares in AL Sydbank by managers and closely associated persons
Globe NewsWire News Distribution Service Article Count: 1523
Media Partner Network Article Count: 4
Send2Press NewsWire Distribution Service Article Count: 532
Related Product Search/Búsqueda de productos relacionados
















